Fluorine »
PDB 5j8m-5jw1 »
5jv0 »
Fluorine in PDB 5jv0: Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038
Enzymatic activity of Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038
All present enzymatic activity of Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038:
2.5.1.1; 2.5.1.10;
2.5.1.1; 2.5.1.10;
Protein crystallography data
The structure of Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038, PDB code: 5jv0
was solved by
J.Park,
C.Y.Leung,
Y.S.Tsantrizos,
A.M.Berghuis,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 41.70 / 2.40 |
| Space group | P 41 21 2 |
| Cell size a, b, c (Å), α, β, γ (°) | 111.540, 111.540, 75.980, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 17.6 / 23.5 |
Fluorine Binding Sites:
The binding sites of Fluorine atom in the Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038
(pdb code 5jv0). This binding sites where shown within
5.0 Angstroms radius around Fluorine atom.
In total 3 binding sites of Fluorine where determined in the Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038, PDB code: 5jv0:
Jump to Fluorine binding site number: 1; 2; 3;
In total 3 binding sites of Fluorine where determined in the Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038, PDB code: 5jv0:
Jump to Fluorine binding site number: 1; 2; 3;
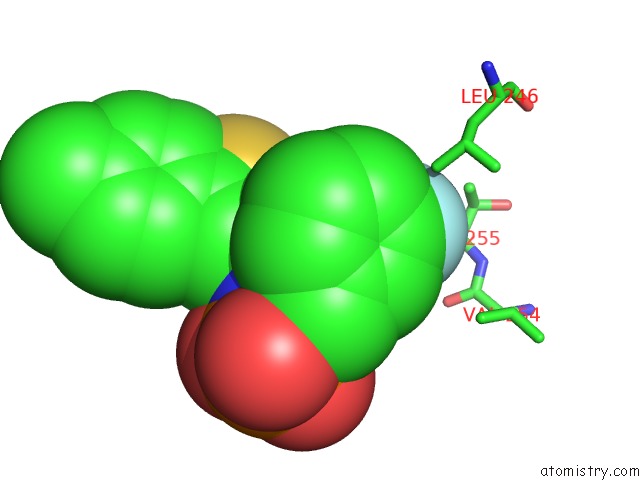
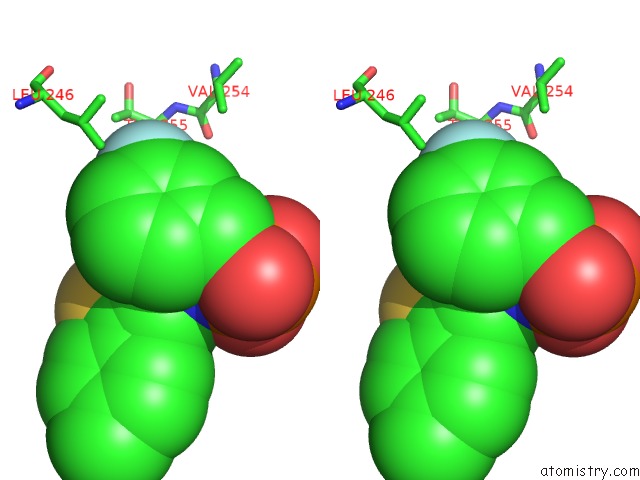
Fluorine binding site 1 out of 3 in 5jv0
Go back to
Fluorine binding site 1 out
of 3 in the Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 1 of Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038 within 5.0Å range:
|
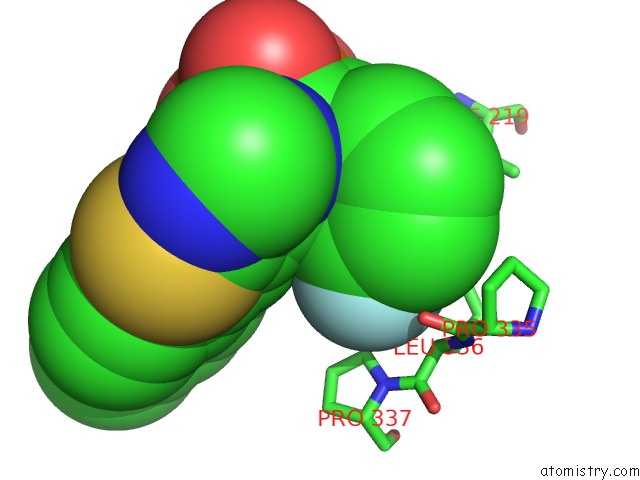
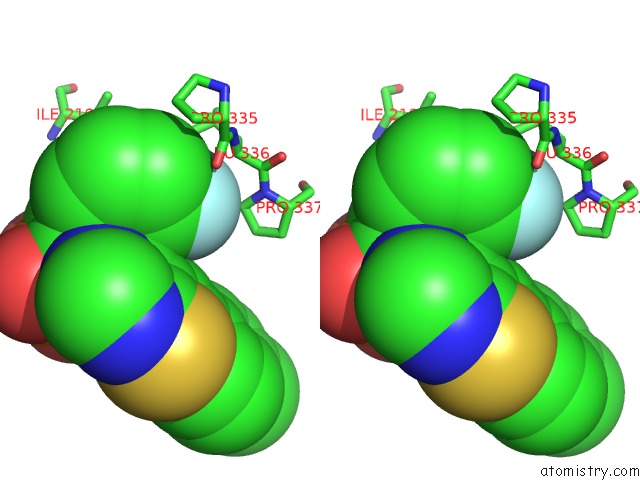
Fluorine binding site 2 out of 3 in 5jv0
Go back to
Fluorine binding site 2 out
of 3 in the Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 2 of Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038 within 5.0Å range:
|
Fluorine binding site 3 out of 3 in 5jv0
Go back to
Fluorine binding site 3 out
of 3 in the Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038
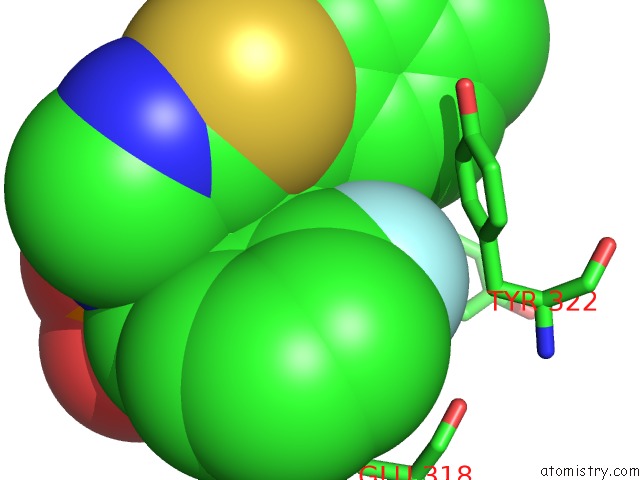
Mono view
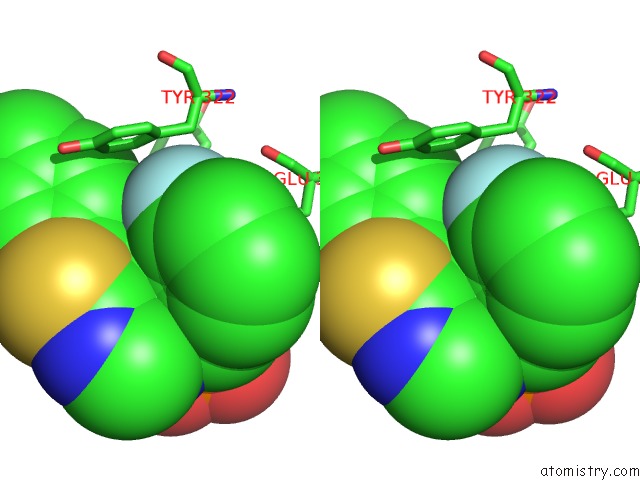
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 3 of Crystal Structure of Human Fpps in Complex with An Allosteric Inhibitor Cl-08-038 within 5.0Å range:
|
Reference:
J.Park,
C.Y.Leung,
A.N.Matralis,
C.M.Lacbay,
M.Tsakos,
G.Fernandez De Troconiz,
A.M.Berghuis,
Y.S.Tsantrizos.
Pharmacophore Mapping of Thienopyrimidine-Based Monophosphonate (Thp-Mp) Inhibitors of the Human Farnesyl Pyrophosphate Synthase. J. Med. Chem. V. 60 2119 2017.
ISSN: ISSN 1520-4804
PubMed: 28208018
DOI: 10.1021/ACS.JMEDCHEM.6B01888
Page generated: Thu Aug 1 10:42:38 2024
ISSN: ISSN 1520-4804
PubMed: 28208018
DOI: 10.1021/ACS.JMEDCHEM.6B01888
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW