Fluorine »
PDB 6gqy-6hay »
6h90 »
Fluorine in PDB 6h90: K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A.
Enzymatic activity of K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A.
All present enzymatic activity of K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A.:
5.4.2.6;
5.4.2.6;
Protein crystallography data
The structure of K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A., PDB code: 6h90
was solved by
A.J.Robertson,
C.Bisson,
J.P.Waltho,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 40.75 / 1.31 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 52.320, 53.630, 81.500, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 13.5 / 16.3 |
Other elements in 6h90:
The structure of K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A. also contains other interesting chemical elements:
| Magnesium | (Mg) | 1 atom |
| Sodium | (Na) | 1 atom |
Fluorine Binding Sites:
The binding sites of Fluorine atom in the K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A.
(pdb code 6h90). This binding sites where shown within
5.0 Angstroms radius around Fluorine atom.
In total 3 binding sites of Fluorine where determined in the K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A., PDB code: 6h90:
Jump to Fluorine binding site number: 1; 2; 3;
In total 3 binding sites of Fluorine where determined in the K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A., PDB code: 6h90:
Jump to Fluorine binding site number: 1; 2; 3;
Fluorine binding site 1 out of 3 in 6h90
Go back to
Fluorine binding site 1 out
of 3 in the K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A.
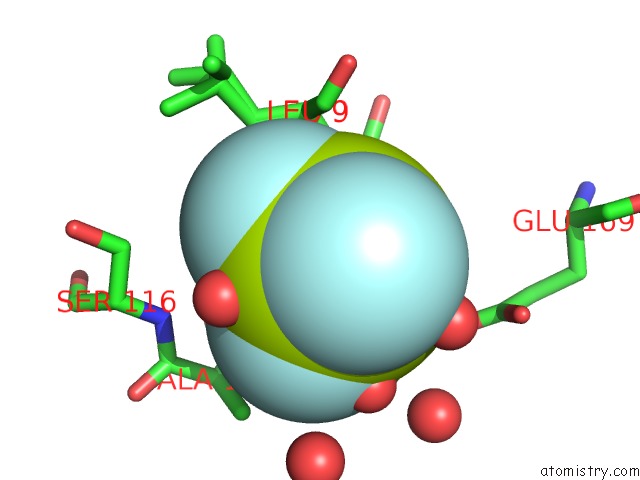
Mono view
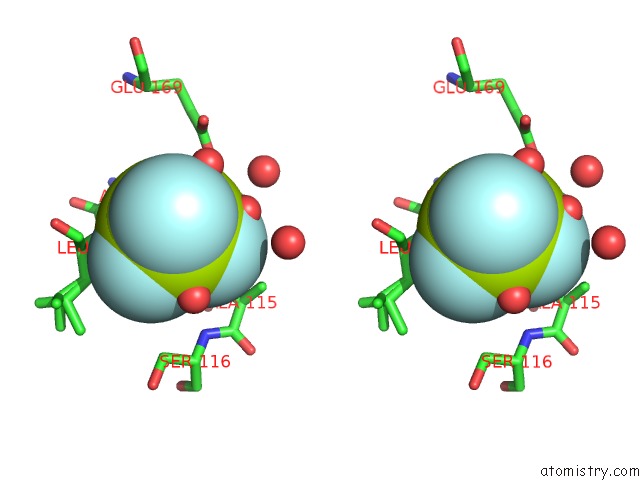
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 1 of K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A. within 5.0Å range:
|
Fluorine binding site 2 out of 3 in 6h90
Go back to
Fluorine binding site 2 out
of 3 in the K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A.
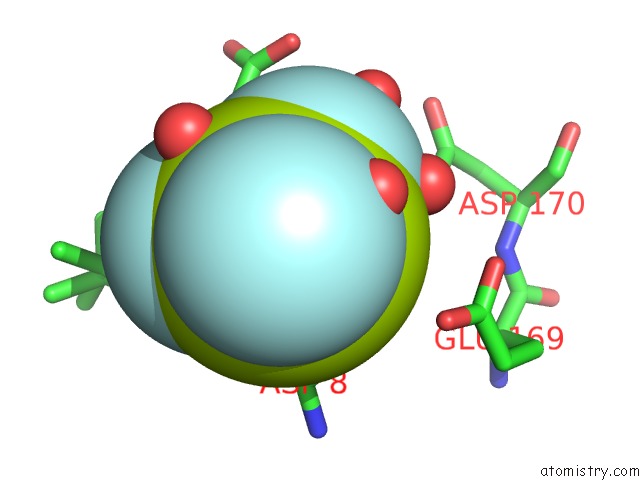
Mono view
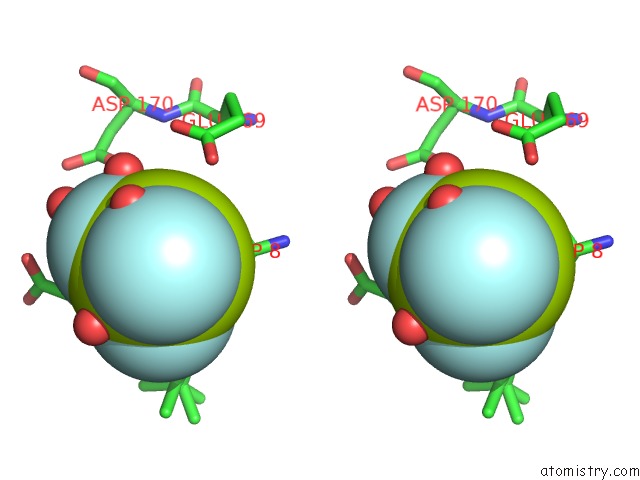
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 2 of K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A. within 5.0Å range:
|
Fluorine binding site 3 out of 3 in 6h90
Go back to
Fluorine binding site 3 out
of 3 in the K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A.
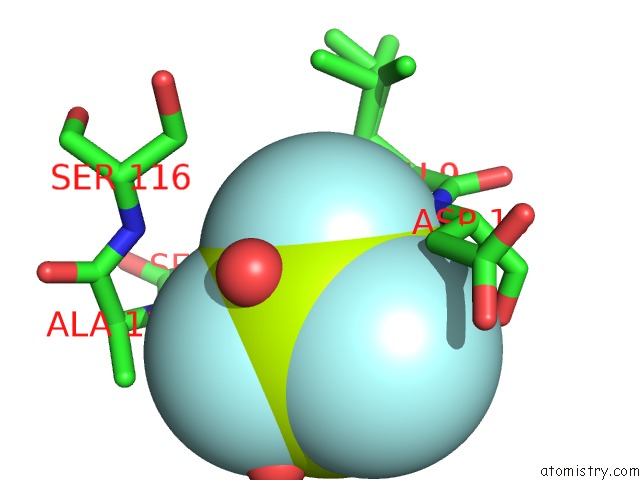
Mono view
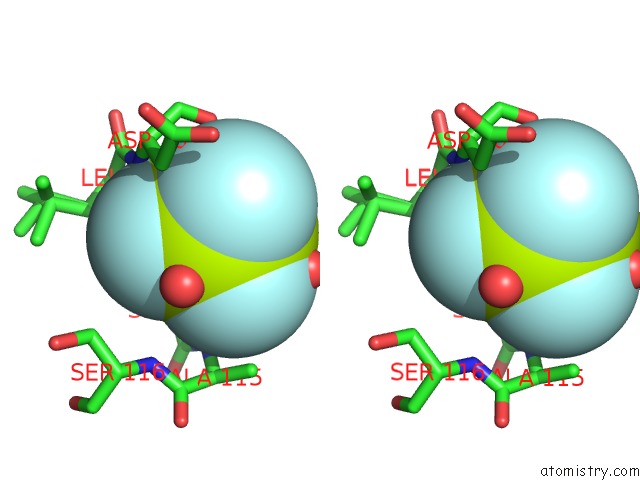
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 3 of K145A Variant of Beta-Phosphoglucomutase From Lactococcus Lactis Inhibited By Beryllium Trifluoride to 1.3 A. within 5.0Å range:
|
Reference:
A.J.Robertson,
C.Bisson,
J.P.Waltho.
Transition State of Phospho-Enzyme Hydrolysis in Beta-Phosphoglucomutase. To Be Published.
Page generated: Tue Jul 15 12:09:28 2025
Last articles
F in 7K0WF in 7JYR
F in 7JY3
F in 7JYQ
F in 7JX9
F in 7JY1
F in 7JY0
F in 7JXW
F in 7JXZ
F in 7JXK