Fluorine »
PDB 5qc1-5qot »
5qdv »
Fluorine in PDB 5qdv: Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A
Enzymatic activity of Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A
All present enzymatic activity of Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A:
3.1.3.48;
3.1.3.48;
Protein crystallography data
The structure of Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A, PDB code: 5qdv
was solved by
D.A.Keedy,
Z.B.Hill,
J.T.Biel,
E.Kang,
T.J.Rettenmaier,
J.Brandao-Neto,
F.Von Delft,
J.A.Wells,
J.S.Fraser,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 44.26 / 1.77 |
| Space group | P 31 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 90.242, 90.242, 107.386, 90.00, 90.00, 120.00 |
| R / Rfree (%) | 19.2 / 21.4 |
Fluorine Binding Sites:
The binding sites of Fluorine atom in the Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A
(pdb code 5qdv). This binding sites where shown within
5.0 Angstroms radius around Fluorine atom.
In total 2 binding sites of Fluorine where determined in the Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A, PDB code: 5qdv:
Jump to Fluorine binding site number: 1; 2;
In total 2 binding sites of Fluorine where determined in the Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A, PDB code: 5qdv:
Jump to Fluorine binding site number: 1; 2;
Fluorine binding site 1 out of 2 in 5qdv
Go back to
Fluorine binding site 1 out
of 2 in the Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A
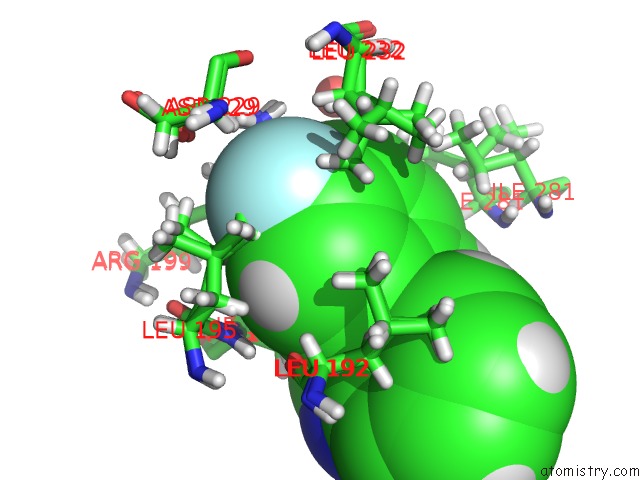
Mono view
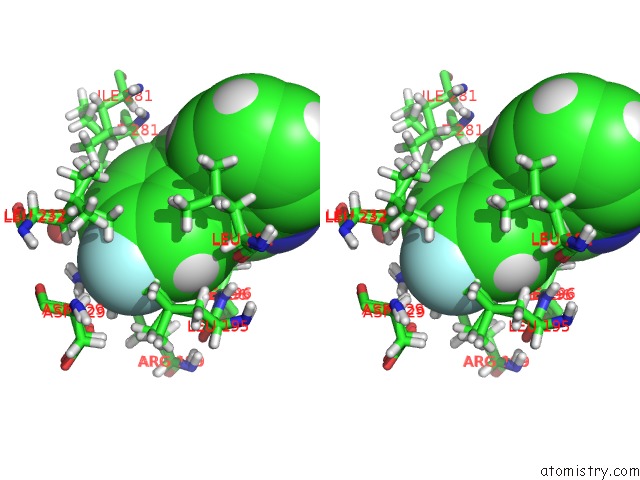
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 1 of Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A within 5.0Å range:
|
Fluorine binding site 2 out of 2 in 5qdv
Go back to
Fluorine binding site 2 out
of 2 in the Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A
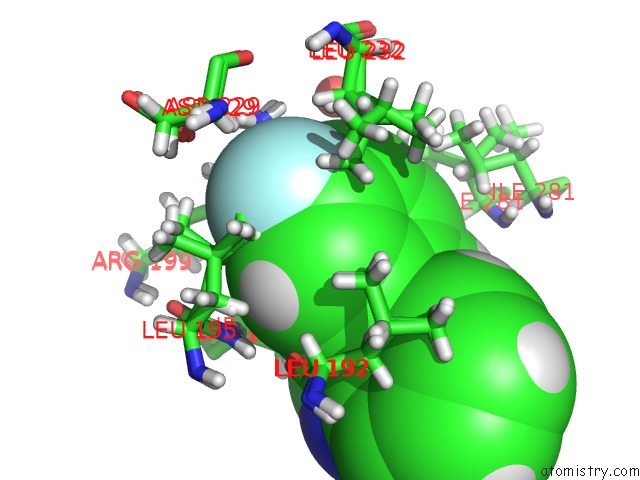
Mono view
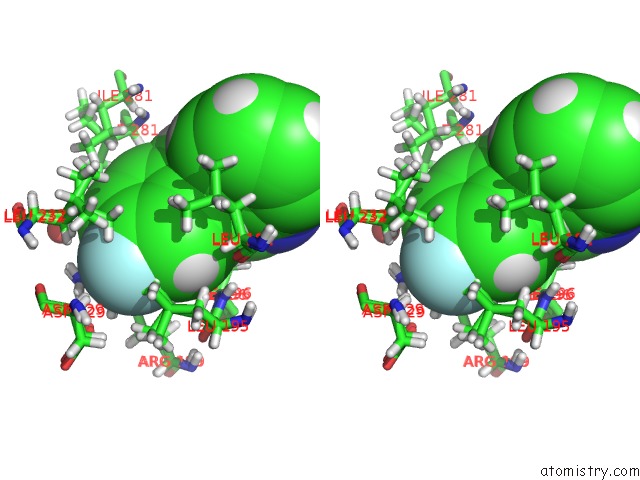
Stereo pair view

Mono view

Stereo pair view
A full contact list of Fluorine with other atoms in the F binding
site number 2 of Pandda Analysis Group Deposition -- Crystal Structure of PTP1B in Complex with COMPOUND_FMOPL000574A within 5.0Å range:
|
Reference:
D.A.Keedy,
Z.B.Hill,
J.T.Biel,
E.Kang,
T.J.Rettenmaier,
J.Brandao-Neto,
N.M.Pearce,
F.Von Delft,
J.A.Wells,
J.S.Fraser.
An Expanded Allosteric Network in PTP1B By Multitemperature Crystallography, Fragment Screening, and Covalent Tethering. Elife V. 7 2018.
ISSN: ESSN 2050-084X
PubMed: 29877794
DOI: 10.7554/ELIFE.36307
Page generated: Tue Jul 15 06:17:41 2025
ISSN: ESSN 2050-084X
PubMed: 29877794
DOI: 10.7554/ELIFE.36307
Last articles
F in 7M0TF in 7M00
F in 7M01
F in 7M03
F in 7M02
F in 7LZU
F in 7LZW
F in 7LY8
F in 7LWG
F in 7LZV